Class 12 Physics Chapter 11 Dual Nature of Radiation and Matter
Photoelectric effect occurs when light of frequency greater than threshold frequency fall on metal plate, electrons get emitted from the metal surface. In this blog we will talk minor details of photoelectric effect.
Story of Photoelectric Effect
Heinrich Hertz, was experimenting on the electromagnetic waves. He tried to produce electromagnetic waves with the help of spark discharge. In his observation he found that, when emitter plate was illuminated by ultraviolet light, the high voltage spark across the detector loop was enhanced. He concluded that, when light fall on the metal surface, some electrons present near the surface absorbs energy from it. However, if the energy is sufficient to overcome the attractive forces of ions present in metal surface, then these electrons escape from the surface into surrounding space.

Photoelectric effect: Lenard’s Experiment
In similar type of experiment, Philipp Lenard also observed that, when ultraviolet light fall on emitter plate of two electrodes present in evacuated tube, causes flow of current through the tube. As soon as the radiation of ultraviolet light on emitter stops the current stops flowing.
Hallwach in 1888, connected a negatively charged zinc plate to an electroscope. He illuminated plate with ultraviolet light. It was found that plate lost its charge, whereas, positively charged plate was found to more positive. He concluded that, negatively charged particles were emitted from the zinc plate under the action of ultraviolet light.
It was the time of discovery of electron. Hence it became evident that incident light causes electrons to emitted from the emitter plate. Due to electric field between the plates, this electron moves towards positive plate. Also, it was found that electron emission was possible only for light of frequency greater than certain minimum frequency striking the plate. This frequency is called as threshold frequency. And thus, photoelectric effect is defined as,
“When light of frequency greater than threshold frequency fall on the metal plate, electrons get emitted from the metal surface.”
Photoelectric effect with different metals
With different metals, photoelectric effect occurs at different conditions. Metals like Zinc, Cadmium, Magnesium, etc, responded only to ultraviolet light, having short wavelength, or high frequency, thus result in electron emission from the surface. However, metals like Lithium, Sodium, Potassium, Caesium and Rubidium show photoelectric effect even for the illumination of visible light.
Experimental study of photoelectric effect
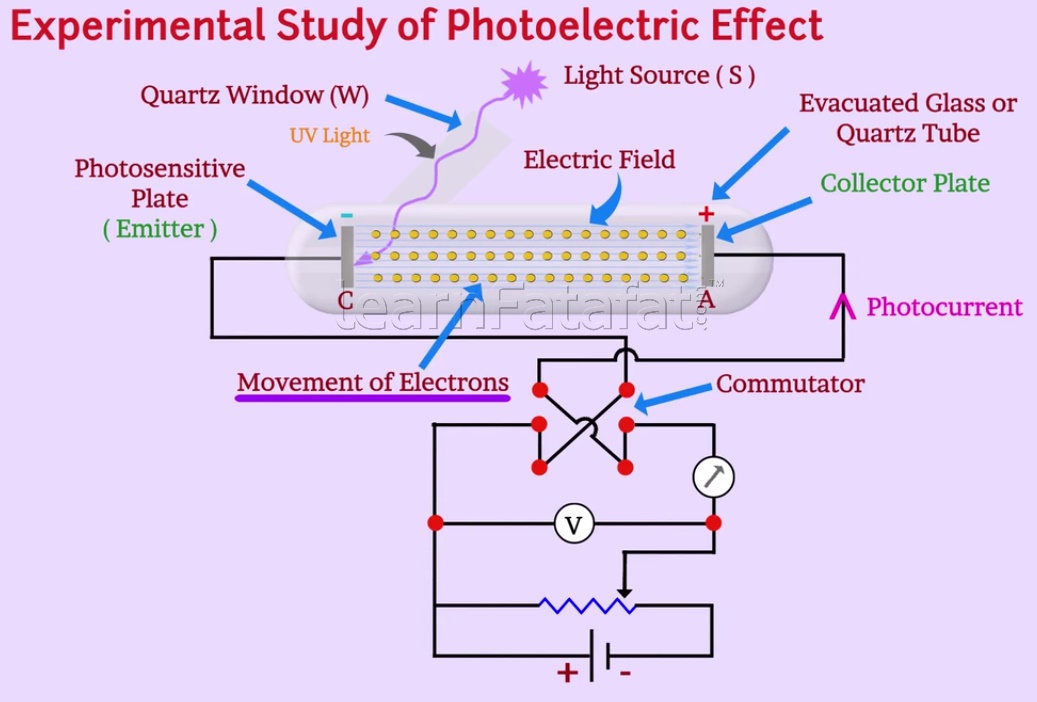
Experimental Study of Photoelectric Effect
Setup contains evacuated glass or quartz tube having a photosensitive plate. Photosensitive plate acts as emitter whereas metal plate acts as a collector. A monochromatic light from the light source is passed through the window, thus light falls on the emitter plate. Window is made up of quartz is sealed on glass tube, moreover, it permits ultraviolet radiations to pass through it. Battery of variable potential difference connected to plates emitter and collector setting up the electric field between them. The electric field between plates helps to enhance movement of emitted electron from emitter plate to collector plate. As battery maintains the potential difference between both the plates, potential difference can be varied. Therefore collector plate can be maintained at desired positive or negative potential with respect to emitter plate. Ultraviolet light falling on the metal plate emit electrons. These electrons moves towards the collector, as the collector is positive. Thus current is set up in the circuit which is known as photocurrent. Variation of photoelectric current can be understood with intensity of radiation, potential difference between emitter and collector, frequency of incident radiation and nature of material of emitter plate.
Definition of Saturation Current and Stopping Potential
Saturation current is maximum value of photoelectric current when all the photoelectrons emitted by the emitted plate are collected by the collector plate.
For particular frequency of incident radiation, the value of negative potential V0 given to the collector plate, at which photocurrent stops or becomes zero is called cutoff or stopping potential.
Wave theory of light and Photoelectric effect
Light has wave nature. Several phenomena like interference, diffraction, polarisation are explained by wave nature of light. This suggest that light is the electromagnetic wave having both electric and magnetic field. Let’s speak about phenomena of photoelectric effect in terms of wave nature of light. Wave theory of light failed to explain the phenomena of photoelectric effect. It was found that, light with smaller frequency and high intensity if allowed to fall on the metal surface, then the photoelectric effect does not occur. Moreover, wave theory states that absorption of energy by electron takes place continuously over entire wavefront of radiation. As large number of electrons absorb energy absorbed per electron per unit time is very small. Further, mathematical calculations showed that it takes hours or more for single electron to absorb sufficient energy and get knocked out from metal surface. But in reality light of sufficient frequency result in electron emission as soon as it strikes over the metal surface. This was contradiction to wave nature of light. Thus wave theory failed to explain basic features of photoelectric effect.
Albert Einstein proposed a completely new picture of electromagnetic radiation to explain photoelectric effect. According, to Einstein phenomena of photoelectric effect is instantaneous, irrespective of intensity. This is because, photon strike over an electron, impart its energy to electron and coming out of electron is almost instantaneous. Intensity only determines number of electrons taking part in the process.
Keywords: Electromagnetic radiation, Photoelectron, Wave theory of light, Monochromatic light
Related articles
Kepler’s Law of Planetary Motion
How does lens in human eye works?
What is surface tension in liquids?
Concept of Polarization of Light
Doppler Effect


Very simple & easy to understand
Thanks to the wonderful guide
I enjoy the article