States of matter exist in three states, solid state, liquid state and gaseous state. States of Matter Solids have definite shape, size and fixed volume. Particles of solids are held with strong force of attraction, hence, there are less vacant spaces between the particles. This makes solids very less compressible. Moreover, solids are rigid and can break if external force is applied. Liquids do not have definite shape, but volume of liquid is fixed. Particles in liquid are held with less force of attraction as compared to solid. Hence, there are more vacant spaces present in liquids as compared to solid. Gases neither have definite shape nor have fixed volume as that of solid and liquid state of matter. Particles in gaseous state of matter have very weak force of attraction, hence, they have large vacant spaces. Therefore, gases are highly compressible. Best example of compressibility of gas can be seen in LPG cylinders used for household purposes. In LPG cylinders, gases are compressed to liquid and then stored in containers.
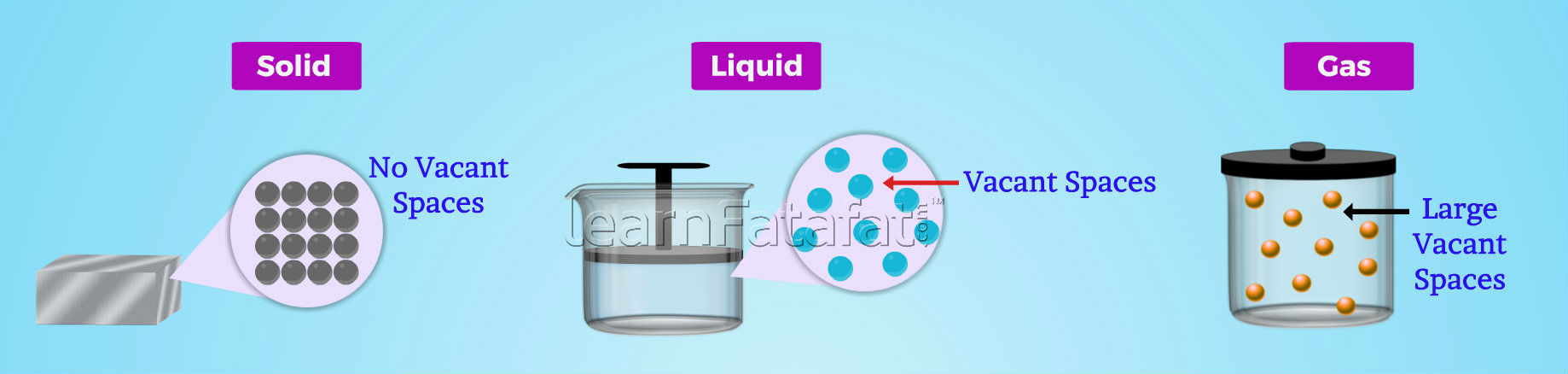
Solid state of matter
Liquid state of matter
Therefore, liquids are more compressible than solids. Liquids do not possess rigidity as that of solidsGaseous state of matter
Comparison between States of matter
Properties
Solid
Liquid
Gas
Shape
Definite Shape
No definite shape
No definite shape
Volume
Fixed volume
Fixed volume
No fixed volume
Space
No vacant spaces between particles
Have small vacant spaces between particles
Have large spaces between the particles
Compressibility
Less compressible
More compressible than solids
Highly compressible
Rigidity
Possess rigidity
Not rigid
Not rigid
Change in state of matter


This article is helpful for Class 9 Science
Keywords: States of matter, Solid, Liquid, Gases, Sublimation, Melting point, Boiling point
Related articles
Reaction Of Metals With Oxygen
Physical Properties Of Metal And Non-metal
Periodic Table And Periodic Trends
Chemistry Of Hydrogen
The Carbon Family


