NCERT Solutions for Class 9 Science
Chapter 1 Matter In Our Surroundings
NCERT Solution for Class 9 Maths and Science (Free). LearnFatafat has created free NCERT Solutions for Class 9 Science Chapter 1 Matter In Our Surroundings. In this Chapter 1 Matter In Our Surroundings, you will learn Physical Nature of Matter and Characteristics of Particles, States of Matter, Can Matter Change its State, Effects of Change in Pressure on States of Matter and Evaporation, Evaporation and Cooling. This class 9 Science Chapter 1 Matter In Our Surroundings course is consists of Notes, M.C.Q’s, Videos and Free NCERT Solutions.
NCERT Solutions for Class 9 Science Chapter 1 Matter In Our Surroundings
1. Convert the following temperatures to the Celsius scale. (a) 300 K (b) 573 K.
Answer: (a) 300 K = (300 – 273) °C = 27 °C
(b) 573 K = (573 – 273) °C = 300 °C
2. Convert the following temperatures to the Kelvin scale. (a) 25°C (b) 373°C.
Answer: (a) 25°C = ( 25 + 273 ) K = 298 K
(b) 373°C = ( 373 + 273 ) K = 646 K
3. Give reason for the following observations.
(a) Naphthalene balls disappear with time without leaving any solid.
Answer: Naphthalene balls are sublimable substance. Thus, naphthalene balls gets converted into gas directly from the solid. Therefore, naphthalene balls disappear with time without leaving solid.
(b) We can get the smell of perfume sitting several metres away.
Answer: Gas particles have high speed and large spaces between them. Perfume particles gets diffuse in these gaseous particles at faster rates thereby reaching our nostrils. Thus we can smell perfume sitting several metres away.
4. Arrange the following substances in increasing order of forces of attraction between the particles— water, sugar, oxygen.
Answer: Forces of attraction in solid is larger than liquids and forces of attraction in liquids in larger than in gases. Accordingly, sugar being solid has greater force of attraction than water and water being liquid has greater force of attraction than oxygen. Therefore, correct increasing order is oxygen less than water less than glucose.
5. What is the physical state of water at— (a) 25°C (b) 0°C (c) 100°C ?
Answer: (a) 25 °C – At this temperature water exist in liquid state.
(b) 0 °C – At this temperature water can exist in solid as well as liquid state. This is because, due to latent heat of fusion, ice starts to melt, which converts it into water.
(c) 100 °C – At this temperature water can exist in liquid as well as gaseous form. This is because, due to latent heat of vaporization, liquids starts to evaporate and convert into water vapors.
6. Give two reasons to justify—
(a) water at room temperature is a liquid.
Answer: Change in state of water takes place from 0°C to 100°C. Thus water is found in liquid state. Also, water has the characteristic to flow at room temperature.
(b) an iron almirah is a solid at room temperature.
Answer: Melting and boiling points of iron is higher around 1500°C. Room temperature is about 20 to 30°C. Thus, iron almirah is solid at room temperature.
7. Why is ice at 273 K more effective in cooling than water at the same temperature?
Answer: In comparison to water ice has less energy even if both are same temperature. Water has more latent heat of fusion as compared to ice. Thus, ice at 273 K is more effective in cooling than water.
8. What produces more severe burns, boiling water or steam?
Answer: Latent heat of vaporization is more in steam as compared to boiling water. Thus steam has more energy. Thus, burns produced by steam is severe than boiling water.
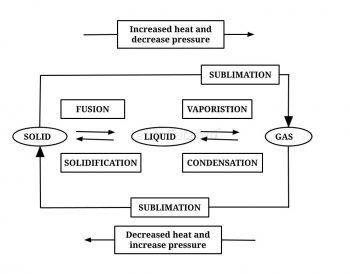
9. Name A,B,C,D,E and F in the following diagram showing change in its state

Answer:
Check other lessons NCERT Solutions for Class 9
Download free NCERT textbook class 9 Science – Chapter 1 Matter In Our Surroundings
CBSE Class 9
Access Class 9 video lessons online (internet required) or offline(internet not required) in SD Card, Pendrive, DVD, Tablet.
Chapter 1 – Matter In Our Surroundings






