- Mixture: Two or more substances mixed with each other and can be distinctly identified is called as mixture. E.g. Mixture of wheat and stone, mixture of rice flakes and raisins.
- Homogeneous mixture: A mixture in which particles of substances are uniformly distributed is called as homogeneous mixture. Homogeneous mixtures can have variable composition. E.g. Mixture of salt and water.
- Heterogeneous mixture: A mixture in which particles of substances are non uniformly distributed is called as heterogeneous mixture. E.g. Mixture of water and stones.
- Solution: A solution is a homogeneous mixture of two or more substances. Solution can be solid solution, liquid solution and gaseous solution.
- Components of solution:
- Solute: Substance in a solution present in less quantity is called as solute.
- Solvent: Substance in a solution present in large quantity is called as solvent.
- E.g. Salt is solute and water is solvent in salty solution.
- Concentration of solution: It is amount of solute present in the solution.
- Dilute solution: Solute is present in small quantity in a solution.
- Concentration of solution: Solute is present in large quantity in solution.
- Formula to calculate concentration of solution:
- Mass by mass percentage of solution = (mass of solute / mass of solution) × 100
- Mass by volume percentage of solution = (mass of solute / volume of solution) × 100
- Saturated solution: Solution dissolves solute to its dissolving capacity.
- Unsaturated solution: Amount of solute in a solution less than the dissolving capacity of solution is called as unsaturated solution.
- Solubility of solution: It is the amount of solute present in the saturated solution.
- Suspension: It is a heterogeneous mixture in which particle of solute do not dissolve and remain suspended within the solution.
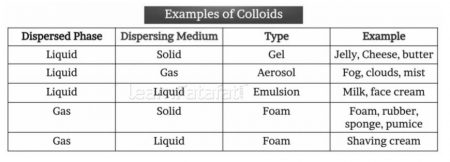
- Colloidal solution: Solution is heterogeneous, in which particle size is larger than solution and smaller than suspension.
- Component of colloid:
- Dispersed phase: Contains solute particles or dispersed particles
- Dispersion medium: Dispersed phase is suspended in dispersion medium
- Comparison between properties of solution, colloid and suspension:
- Tyndall effect: It is the scattering of light due to solute particles in colloids or in suspension.
- Techniques for separation of components of mixture:
- Used in dairy for separation of butter from cream
- Used in laboratories for blood and urine test
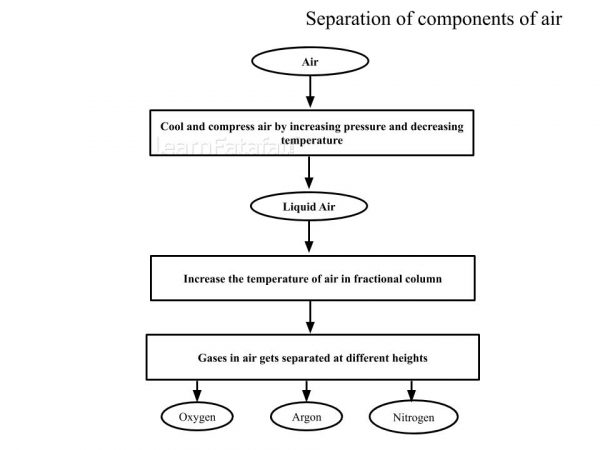
- Separation of components of air: Air is homogeneous mixture of different types of gases. Gases from air are separated by fractional distillation. Steps for separating of air
- Crystallisation: Crystals of pure solid is obtained from the liquid by heating and cooling.
- Pure substances: Pure substances are classified as element and compounds. Elements are further classified into metals, nonmetals and metalloids.
- Metals vs nonmetals:
- Metalloids: Show properties in between metals and nonmetals. E.g. Germanium, Silicon, Arsenic, etc.
- Compound: It is formed when two or more elements chemically combine with each other, and forms new substance with new properties.
- Distinguish between mixtures and compounds:
- Overlook of mixtures and pure substances:
| Solution | Colloid | Suspension | |
|---|---|---|---|
| Particle size | Very small | Larger than solution but smaller than suspension | Larger |
| Visibility of particle | Not visible by naked eye | Not visible by naked eye | Visible by naked eye |
| Tyndall effect | Do not scatter beam of light | Scatters beam of light | Scatters beam of light |
| Sedimentation | Particles do not settle down if kept undisturbed | Particles do not settle down if kept undisturbed | Particles settle down if kept undisturbed |
| Separation | Particles cannot be separated by filtration | Particles cannot be separated by filtration but can be separated by centrifugation | Particles are separated by filtration |
| Name | Process | Application(s) |
|---|---|---|
| Evaporation | Separates volatile component from non volatile component. Volatile component evaporates leaving non volatile component | Separation of dye from ink |
| Centrifugation | Separates fine solute particles which from liquid which are not separated through filtration |
|
| Separating funnel | Separates liquids which do not mix with each other | Separation of oil and water |
| Sublimation | Separate sublimable volatile impurity from non sublimable volatile impurity | Separation of camphor from salt |
| Chromatography | Components of mixture are separated using filter paper and solvent. More soluble component rises high on filter paper. | Separation of colors from dye |
| Distillation | Separates mixture of two or more liquids | Separation of ethanol and water |
| Fractional distillation | Separates mixture of liquids having less than 25K difference in boiling point. | Separation of liquids from crude oil |
| Metals | Nonmetals |
|---|---|
| Have lustre | Do not have lustre |
| Good conductors of heat and electricity | Bad conductors of heat and electricity |
| Are ductile, malleable and sonorous | Are not ductile, malleable and sonorous |
| Are hard | Are brittle |
| Are solid in state except mercury | Are in solid, liquid and gaseous state |
| E.g. Iron, Gold, Silver, Sodium, Copper,etc | E.g. Carbon, Hydrogen, Bromine, Oxygen, etc |
| Mixtures | Compounds |
|---|---|
| Elements or compounds are mixed together to form a mixture. No new compound is formed. | Elements react with each other to form new compounds with new properties. |
| Mixtures have variable composition | Compounds have fixed proportion |
| Mixture show original properties of constituents present in it. | Compound do not show original properties of substance of which it is composed. |
| Constituents of mixture are separated by physical methods | Constituents of compounds are separated by chemical process |
| Example : Mixture of Copper filings and sulphur, salt and sand, etc. | Example : Copper Sulphate, Carbon Disulphide, etc. |







